Injury recovery: why you're healing slowly and what biomarkers to check
Slow injury recovery is one of the most frustrating experiences for anyone who trains, competes, or simply wants to stay active. When injury recovery takes longer than expected, the instinct is usually to rest more or train differently. But in many cases, the answer lies in biology: the nutrients, hormones, and inflammatory markers that govern how efficiently your body repairs damaged tissue. Understanding what is driving your slow healing specifically requires looking at the biomarkers most commonly involved, and this is where targeted testing becomes useful.
What causes slow injury recovery?
Vitamin D deficiency
Vitamin D is the most consistently studied nutrient in relation to injury healing and recovery. Skeletal muscle tissue contains specific vitamin D receptors, and the evidence points to a direct role in muscle cell regeneration, collagen production, and immune modulation during the repair process. Studies show a positive correlation between serum vitamin D levels and strength recovery after muscle injury. Research in athletes has also found that vitamin D deficiency is common in both recreational and professional athletes, and that deficiency increases injury risk and slows the adaptive response to exercise. A vitamin D level below 50 nmol/L is considered insufficient; many sports medicine practitioners target levels above 100 nmol/L for optimal musculoskeletal recovery.
Low ferritin and iron status
Iron deficiency is one of the most common and most overlooked contributors to slow injury recovery. Ferritin, the storage form of iron, is required for the enzymatic process that converts T4 into active T3 in thyroid hormone metabolism, but its relevance to injury healing is more direct: iron is essential for collagen synthesis, immune cell function, and oxygen transport to healing tissue. Low ferritin impairs the proliferation of fibroblasts, the cells responsible for laying down new connective tissue at injury sites. A person can have ferritin in the technically normal range and still experience significantly impaired healing capacity, particularly if levels are below 30 ng/mL.
Elevated inflammation (CRP and chronic systemic stress)
Acute inflammation is a necessary part of the healing process. The problem arises when inflammation becomes chronic and low-grade, persistently elevated without the acute injury signal that would normally resolve it. High-sensitivity CRP (C-reactive protein) is a marker of systemic inflammation. Chronically elevated CRP suppresses the proliferative and remodelling phases of tissue repair, extending recovery timelines and reducing the quality of healed tissue. Common contributors to elevated CRP in otherwise healthy people include poor sleep, excess body fat, high stress, gut dysbiosis, and nutrient insufficiencies that the person is unaware of.
Blood glucose and insulin dysregulation
Elevated blood glucose impairs healing by multiple mechanisms: it reduces neutrophil and macrophage function (the immune cells that clear debris at injury sites), stiffens collagen, and reduces blood flow to peripheral tissues. Even in people without diagnosed diabetes, HbA1c in the high-normal range or post-meal glucose excursions above 140 mg/dL can meaningfully slow tissue repair. This mechanism explains why people who feel broadly healthy but eat a highly processed diet often experience noticeably slower recovery from training or minor injuries than expected. Tracking HbA1c alongside healing timelines can reveal metabolic contributors that are otherwise invisible.
Nutrient co-factors: zinc, B12, and homocysteine
Zinc activates over 300 enzymes in the body, many of which are directly involved in DNA synthesis, cell division, and protein synthesis. These processes are the building blocks of tissue regeneration. Zinc deficiency is one of the most common micronutrient deficiencies globally, and it impairs wound healing across both the inflammatory and proliferative phases. Vitamin B12 deficiency contributes to slower healing by impairing cell replication and DNA repair, both of which are required at high rates in injured tissue. Elevated homocysteine, which occurs when B12 and folate metabolism are compromised, creates a pro-inflammatory environment that can interfere with collagen cross-linking and vascular repair.
Thyroid function and metabolic rate
Thyroid hormones regulate the speed of virtually every metabolic process, including tissue repair. Subclinical hypothyroidism, where TSH is elevated but symptoms are vague, is associated with slower collagen synthesis and reduced immune activity. Many people experiencing persistent slow recovery have never had a thyroid panel beyond TSH. Testing Free T3, Free T4, and Anti-TPO antibodies alongside nutritional co-factors gives a complete picture of whether thyroid function is contributing to recovery timelines that are longer than expected for the nature of the injury.
How to test for slow injury recovery
The NHS does not offer a single panel designed for recovery investigation. Most GP assessments will test CRP and perhaps haemoglobin, but the full picture requires covering several systems simultaneously: inflammatory status, nutritional adequacy, metabolic health, and thyroid function.
A comprehensive blood panel for slow injury recovery should include:
Vitamin D (25-OH) shows whether the key nutrient for muscle repair and immune modulation is at an adequate level. Deficiency is extremely common in the UK, particularly in athletes who train indoors or during winter months.
Ferritin reflects iron stores. This is frequently missed from standard panels and is one of the most common nutritional contributors to impaired recovery in both men and women.
CRP (high-sensitivity) reveals the degree of background systemic inflammation that may be interfering with the healing process.
HbA1c reflects average blood glucose over the preceding three months. Even borderline elevations are associated with measurably impaired tissue repair.
Vitamin B12 supports cell replication and DNA synthesis, both essential for tissue regeneration. Deficiency is underdiagnosed, particularly in people following plant-based diets.
Homocysteine measures the by-product of B12 and folate metabolism. Elevated levels create a pro-inflammatory environment and indicate impaired methylation capacity, which affects tissue repair at a cellular level.
TSH and Free T3 assess whether thyroid function is contributing to a globally slowed metabolism that extends recovery timelines.
LDL cholesterol and total lipids may reveal metabolic patterns associated with impaired microvascular supply to healing tissue.
Home blood testing is most useful for people whose injuries are healing more slowly than expected and who want to identify whether a nutritional or metabolic factor is involved, rather than simply waiting it out. It is also valuable for athletes who want to optimise their recovery baseline between competitions or training blocks.
Evidence-based strategies to support injury recovery
Prioritise protein and collagen-supporting nutrients
Protein is the most important dietary factor in tissue repair. It provides the amino acids required for collagen production, immune cell synthesis, and muscle fibre regeneration. Research suggests most people need considerably more protein during injury recovery than normal daily activity requires. Alongside adequate protein, vitamin C plays a specific role in collagen cross-linking and is often underestimated in recovery nutrition plans. Good sources include citrus fruits, kiwi, bell peppers, and broccoli. Tracking these alongside a biomarker panel that includes ferritin and B12 tells you whether your dietary intake is translating into functional nutritional status.
Address background inflammation through diet and sleep
Chronic low-grade inflammation is addressable through consistent, compounding lifestyle choices. Omega-3 fatty acids (from oily fish, walnuts, flaxseed) reduce the production of pro-inflammatory prostaglandins and have been shown to support soft tissue healing after injury. Reducing processed food consumption, which drives omega-6 dominance and inflammation, is equally important. Sleep quality is a non-negotiable factor: growth hormone, which drives tissue repair, is predominantly secreted during deep sleep. Tracking CRP over time gives an objective measure of whether lifestyle changes are actually reducing systemic inflammation.
Optimise vitamin D through testing, not guessing
The UK climate makes vitamin D insufficiency the default state for most people from October to April, and for many year-round. Supplementing without testing is unpredictable: some people absorb well at low doses, others require much higher levels to reach the ranges associated with optimal musculoskeletal function. Testing vitamin D before and after supplementation removes the guesswork. Studies consistently find that vitamin D deficiency prolongs recovery from both muscle injury and stress fractures, and that correcting deficiency measurably improves the speed and quality of repair.
Manage blood glucose around training and recovery
Moderating post-meal glucose excursions during a recovery period is one of the most underused levers in recovery nutrition. Stable blood glucose supports consistent immune function and reduces the glycation of collagen, which impairs the structural quality of repaired tissue. Practical strategies include prioritising protein and fibre early in meals, reducing refined carbohydrate intake during periods of reduced training load, and tracking HbA1c as a measure of whether metabolic patterns are working in favour of healing.
Stride tests that can help with Injury recovery
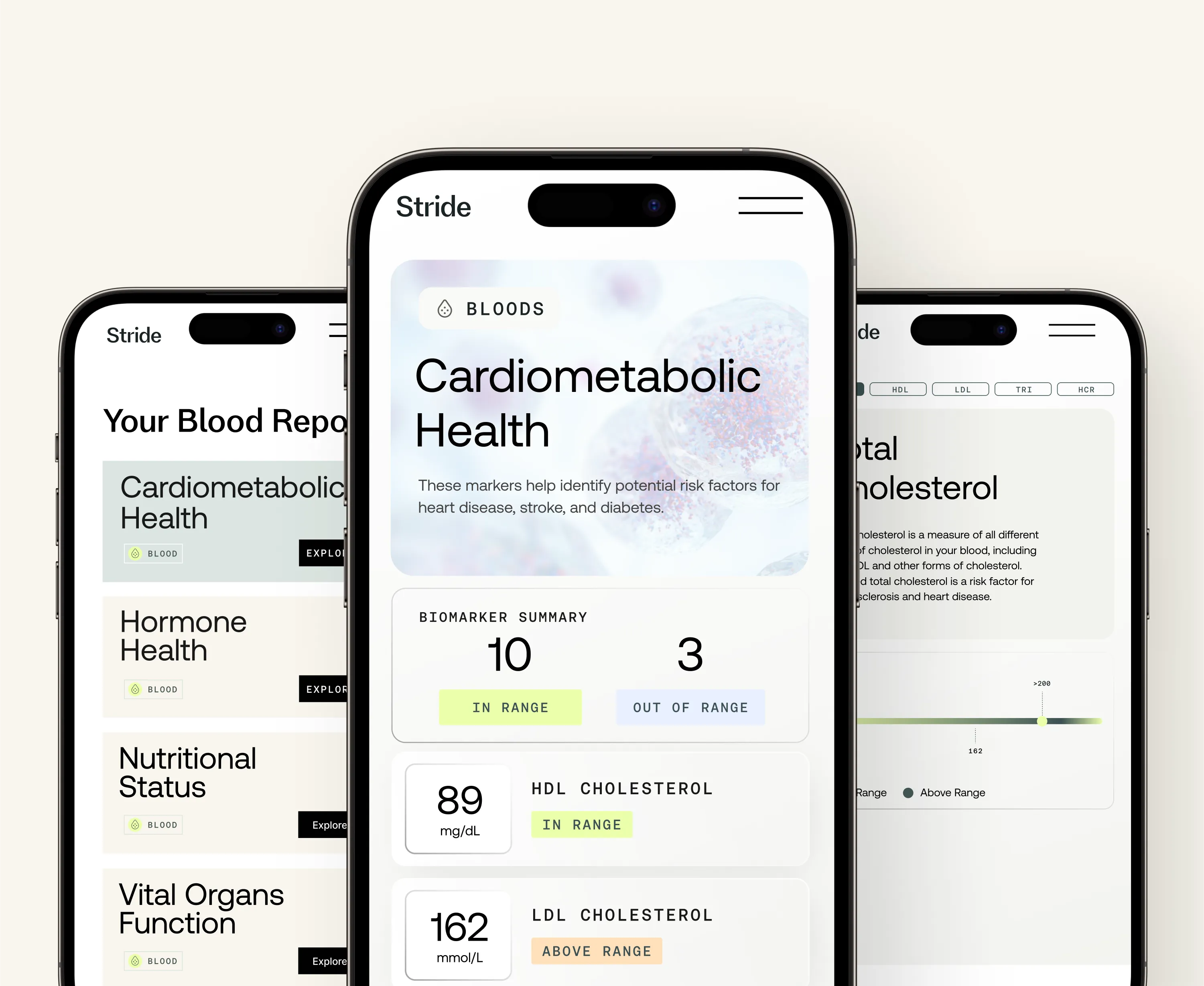
Health Tests
Optimal Bloods
At-home test for more than 70 blood-based biomarkers
From £399 £279.30
Biomarkers
| Biomarker | What it measures | Why it matters | Relevance |
|---|---|---|---|
| Vitamin D Blood Test (25-OH) | 25-OH vitamin D status | Essential for muscle cell regeneration and immune modulation during repair | 5 |
| Ferritin Blood Test | Iron storage levels | Required for collagen synthesis, oxygen transport to healing tissue, and fibroblast function | 5 |
| hsCRP Blood Test (High Sensitivity C-Reactive Protein) | Systemic inflammation marker | Chronically elevated CRP suppresses the proliferative and remodelling phases of healing | 5 |
| HbA1c Blood Test (Glycated Haemoglobin) | Average blood glucose over 3 months | Even borderline elevations impair immune cell function, collagen quality, and microvascular supply | 4 |
| Active B12 Blood Test (Holotranscobalamin) | Active B12 status | Supports cell replication and DNA synthesis required for tissue regeneration | 4 |
| TSH Blood Test (Thyroid Stimulating Hormone) | Pituitary signal for thyroid function | Thyroid status governs the speed of all metabolic processes including tissue repair | 3 |
| FT3 Blood Test (Free Triiodothyronine) | Active thyroid hormone at tissue level | Low T3 slows collagen synthesis and immune activity even when TSH appears normal | 3 |
| LDL Cholesterol Blood Test | Low-density lipoprotein | Elevated LDL patterns can indicate metabolic dysfunction affecting tissue blood supply | 2 |
FAQs
What blood tests should I get if my injuries are healing slowly?
A blood panel for slow injury recovery should cover four key areas: nutritional status (ferritin, vitamin D, vitamin B12, zinc), inflammation (high-sensitivity CRP), metabolic health (HbA1c, fasting glucose), and thyroid function (TSH, Free T3). Standard NHS testing rarely covers all of these in a single draw. Ferritin and vitamin D are the two most commonly deficient nutrients in people with impaired healing and are almost never tested together alongside inflammation markers. A comprehensive panel gives you the full picture rather than chasing one variable at a time, and a consultation with a practitioner who can interpret the interaction between markers is considerably more useful than chasing individual results in isolation.
Can low vitamin D cause slow healing after injury?
Yes, vitamin D deficiency is one of the most well-evidenced nutritional contributors to impaired recovery from musculoskeletal injury. Research shows that serum vitamin D levels positively correlate with strength recovery after muscle injury, and that deficiency reduces the body's ability to regenerate muscle cells and modulate the immune response at the injury site. In the UK, insufficiency is extremely common year-round and near-universal during winter months. Athletes and active people who train indoors or wear high sun protection are at particular risk. Testing and correcting vitamin D deficiency, rather than supplementing blindly, ensures you reach the levels associated with optimal musculoskeletal function rather than simply avoiding clinical deficiency.
Does inflammation affect how quickly you recover from injury?
Acute inflammation is a necessary and beneficial part of the healing process: it clears debris, signals repair, and triggers the arrival of growth factors at the injury site. The problem is chronic, low-grade inflammation that persists outside of an acute injury event. Chronically elevated CRP suppresses the later phases of healing, specifically proliferation (new tissue formation) and remodelling (the organisation and strengthening of repaired tissue). This means the body is stuck in a perpetual early-healing state rather than progressing through the full repair cycle. Common sources of chronic inflammation include poor sleep quality, gut dysbiosis, blood glucose instability, excess body fat, and nutritional deficiencies. Measuring CRP gives an objective baseline and a way to track whether lifestyle changes are actually reducing the inflammatory background.
Can blood sugar affect injury healing?
Yes. Even in people without diabetes, elevated blood glucose patterns impair multiple aspects of injury healing. High glucose reduces the function of neutrophils and macrophages, the immune cells responsible for clearing debris at injury sites and signalling the repair process. It also causes glycation of collagen, which reduces the structural quality of healed tissue and makes it more prone to re-injury. Glucose-driven impairment of microvascular blood flow reduces the delivery of oxygen and nutrients to healing tissue. HbA1c provides a three-month average blood glucose picture and can reveal whether metabolic patterns are working against recovery without any single symptom that would prompt investigation. Tracking HbA1c alongside recovery timelines is particularly relevant for people who eat a highly processed diet or who have sedentary periods during injury rehab.
Why do sports injuries take so long to heal in some people?
Persistent slow healing is almost always multifactorial. The most common contributing factors, in order of prevalence, are: vitamin D insufficiency, low ferritin or iron stores, chronic low-grade inflammation (elevated CRP), blood glucose instability, and subclinical thyroid dysfunction. In athletes, the combination of high training load, inadequate dietary intake during recovery, and reduced sunlight exposure creates a perfect environment for all of these factors to occur simultaneously. Testing all relevant biomarkers in a single draw is considerably more efficient than testing one factor at a time, and it allows you to see how the variables interact. For example, low ferritin amplifies the impact of vitamin D insufficiency, and both affect the immune function that drives tissue repair.
What nutrients are most important for injury recovery?
The nutrients with the strongest evidence for injury recovery are: protein (the raw material for collagen synthesis and muscle regeneration), vitamin D (muscle cell regeneration and immune modulation), vitamin C (collagen cross-linking and connective tissue repair), zinc (cell division, DNA synthesis, and protein turnover), iron/ferritin (collagen synthesis and oxygen transport), and omega-3 fatty acids (anti-inflammatory support during the healing cascade). Crucially, the importance of each nutrient depends on your individual starting levels. Supplementing zinc or vitamin D on top of already adequate levels adds little benefit, while correcting a genuine deficiency in either can dramatically change recovery timelines. A blood panel tells you which of these factors is actually a bottleneck for your specific biology.
Can gut health affect injury recovery?
Gut health affects injury recovery through several mechanisms that are less obvious than direct nutritional intake. The gut microbiome is responsible for approximately 20% of active thyroid hormone conversion (T4 to T3), so gut dysbiosis can impair the thyroid function that governs overall metabolic rate and tissue repair speed. A compromised gut lining also reduces the absorption efficiency of key recovery nutrients including iron, zinc, and B12. Additionally, gut bacteria play a regulatory role in systemic inflammation: dysbiosis creates a low-grade inflammatory background that competes with the body's capacity to mount a focused healing response. If you experience slow recovery alongside digestive symptoms such as bloating or irregular bowel habits, considering both gut microbiome testing and a blood biomarker panel gives a more complete picture.
How long should it take to recover from a muscle injury?
Minor muscle strains typically resolve within one to two weeks. Moderate strains affecting a larger portion of the muscle belly may take four to six weeks. Severe muscle injuries involving significant tissue disruption can take three to six months for full functional recovery. Recovery from tendon injuries is generally slower due to lower blood supply: minor tendinopathy may take six to twelve weeks, while significant tendon damage can take six months or longer. When recovery is taking substantially longer than these typical timeframes, an underlying biological factor is usually involved. Checking the relevant biomarkers, particularly vitamin D, ferritin, and CRP, often reveals a correctable issue. Tracking these biomarkers before injury and monitoring them during recovery also allows you to see whether your interventions are actually moving the relevant markers in the right direction.
Does stress slow down injury healing?
Chronic stress elevates cortisol, which has a measurably negative effect on injury recovery through several pathways. Elevated cortisol suppresses immune function, reducing the effectiveness of the inflammatory response that initiates healing. It also impairs protein synthesis, which is the primary mechanism by which damaged tissue is rebuilt. Research suggests that chronically high cortisol can slow wound healing by up to 40%. Additionally, stress elevates blood glucose, compounding the glycation and microvascular impairment described above. Measuring CRP alongside cortisol-influenced markers such as HbA1c and testing at multiple points over time gives you objective evidence of whether stress is affecting your recovery biology, and a benchmark against which to measure the effect of stress-management interventions.
Find out what your body is actually trying to tell you.
Answer 8 quick questions and get a free personalised report - covering which area of your biology is most worth looking at first, what testing in that area reveals, and what actions become available once you have that data.
Where do you feel you're operating below your potential right now?
Methylation & Cellular Health
Your biology is signalling a methylation upgrade - here’s what that means and what to do about it.










