IBS symptoms: what's causing your gut issues and which tests can help
IBS symptoms affect an estimated 10 to 20 per cent of the global population, making irritable bowel syndrome one of the most common chronic conditions in the UK. Yet despite its prevalence, IBS remains a diagnosis of exclusion: it is confirmed by ruling out other conditions rather than by identifying a specific biological marker. This matters because many people receive an IBS diagnosis without investigating the underlying drivers, including gut microbiome dysbiosis, food sensitivities, thyroid dysfunction, coeliac disease, and iron deficiency anaemia, all of which can produce identical or overlapping symptoms. Understanding what is driving your gut symptoms specifically requires looking at the biomarkers and microbiome patterns most commonly involved, and this is where targeted testing becomes genuinely useful.
What causes IBS and what goes wrong in the gut?
Gut microbiome dysbiosis
Research consistently shows that people with IBS have measurably different gut microbiome compositions compared to healthy controls, characterised by reduced bacterial diversity and shifts in the relative abundance of key species. A 2024 cross-cohort metagenomic study covering more than 9,000 samples identified a reproducible IBS-associated microbial signature for the first time, suggesting that dysbiosis is not merely a correlate of IBS but a likely mechanistic driver. Lower levels of butyrate-producing bacteria (particularly Faecalibacterium prausnitzii and Roseburia) are consistently found in IBS, reducing the short-chain fatty acid production that maintains gut barrier integrity, modulates immune responses, and regulates gut motility.
Altered gut-brain signalling
IBS is classified by gastroenterologists as a disorder of gut-brain interaction (DGBI). The enteric nervous system in the gut contains more neurons than the spinal cord, and its signalling is intimately linked to the central nervous system through the vagus nerve, serotonin pathways, and immune system intermediaries. People with IBS show increased visceral hypersensitivity, meaning the gut's pain threshold is lowered even when the gut lining itself shows no structural abnormality. Serotonin, 90 per cent of which is produced in the gut and directly influenced by gut bacteria, plays a central role in regulating gut motility, and disruptions in serotonin signalling explain many of the motility differences between IBS subtypes (IBS-D, predominantly diarrhoea; IBS-C, predominantly constipation; IBS-M, mixed).
Post-infectious IBS
A significant proportion of people develop IBS following a gastrointestinal infection such as gastroenteritis or food poisoning. Post-infectious IBS (PI-IBS) occurs when the acute inflammatory response of an infection disrupts the gut microbiome in a way that persists beyond recovery. Studies suggest that between 10 and 30 per cent of people who experience a gut infection go on to develop PI-IBS, with risk factors including the severity of the original infection, female sex, and the presence of anxiety or depression at the time of infection.
Food sensitivities and fermentable carbohydrates
IBS symptoms are strongly influenced by diet, particularly the intake of fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAPs). These carbohydrates are incompletely absorbed in the small intestine and rapidly fermented by gut bacteria in the large bowel, producing gas and drawing water into the colon. The degree to which FODMAPs trigger symptoms depends heavily on individual microbiome composition: different bacteria ferment different FODMAP components at different rates, which is why the same dietary trigger produces severe symptoms in one person and none in another with identical food intake.
Stress, anxiety, and psychological factors
IBS and anxiety are bidirectionally linked through the gut-brain axis. People with IBS have elevated rates of anxiety and depression, and people with anxiety have elevated rates of IBS. Stress directly worsens IBS symptoms by altering gut motility, increasing gut permeability, and shifting the microbiome toward dysbiotic compositions. This is not a suggestion that IBS is "all in the head": the psychological and biological components are mechanistically interconnected, and addressing both simultaneously produces better outcomes than treating either in isolation.
Small intestinal bacterial overgrowth (SIBO)
SIBO occurs when bacteria colonise the small intestine in excessive numbers, a region that normally has comparatively few bacteria. An estimated 10 to 15 per cent of the general population may have SIBO, and prevalence among IBS patients may be as high as 80 per cent in some studies, though estimates vary considerably across research methods. SIBO produces IBS-like symptoms including bloating, gas, abdominal pain, and irregular bowel movements through bacterial fermentation of carbohydrates in the small intestine before they can be absorbed. A hydrogen and methane breath test is the standard assessment for SIBO, and the condition is distinct from large bowel dysbiosis measured by standard microbiome testing.
Undiagnosed conditions that cause IBS-like symptoms
Several conditions produce symptoms identical to IBS but require different management: coeliac disease (immune-mediated damage to the small intestinal lining triggered by gluten), inflammatory bowel disease (Crohn's disease and ulcerative colitis), microscopic colitis, bile acid malabsorption, hypothyroidism (which slows gut motility and causes constipation), and iron deficiency anaemia (which causes fatigue and altered gut function). Ruling these out before accepting an IBS diagnosis ensures that a treatable condition is not missed and that dietary interventions are not applied to a condition that requires medical treatment.
How to test for IBS and its underlying causes
There is currently no definitive biomarker test for IBS itself. IBS is diagnosed clinically using the Rome IV criteria, which require recurrent abdominal pain at least once per week for the previous three months, associated with two or more of: pain related to defecation, change in stool frequency, or change in stool form. Blood and stool tests are used to exclude other diagnoses rather than to confirm IBS.
The most useful testing approach for someone with IBS symptoms covers two complementary areas: blood markers that exclude other diagnoses and provide metabolic context, and gut microbiome testing that assesses the specific bacterial patterns associated with the symptom profile.
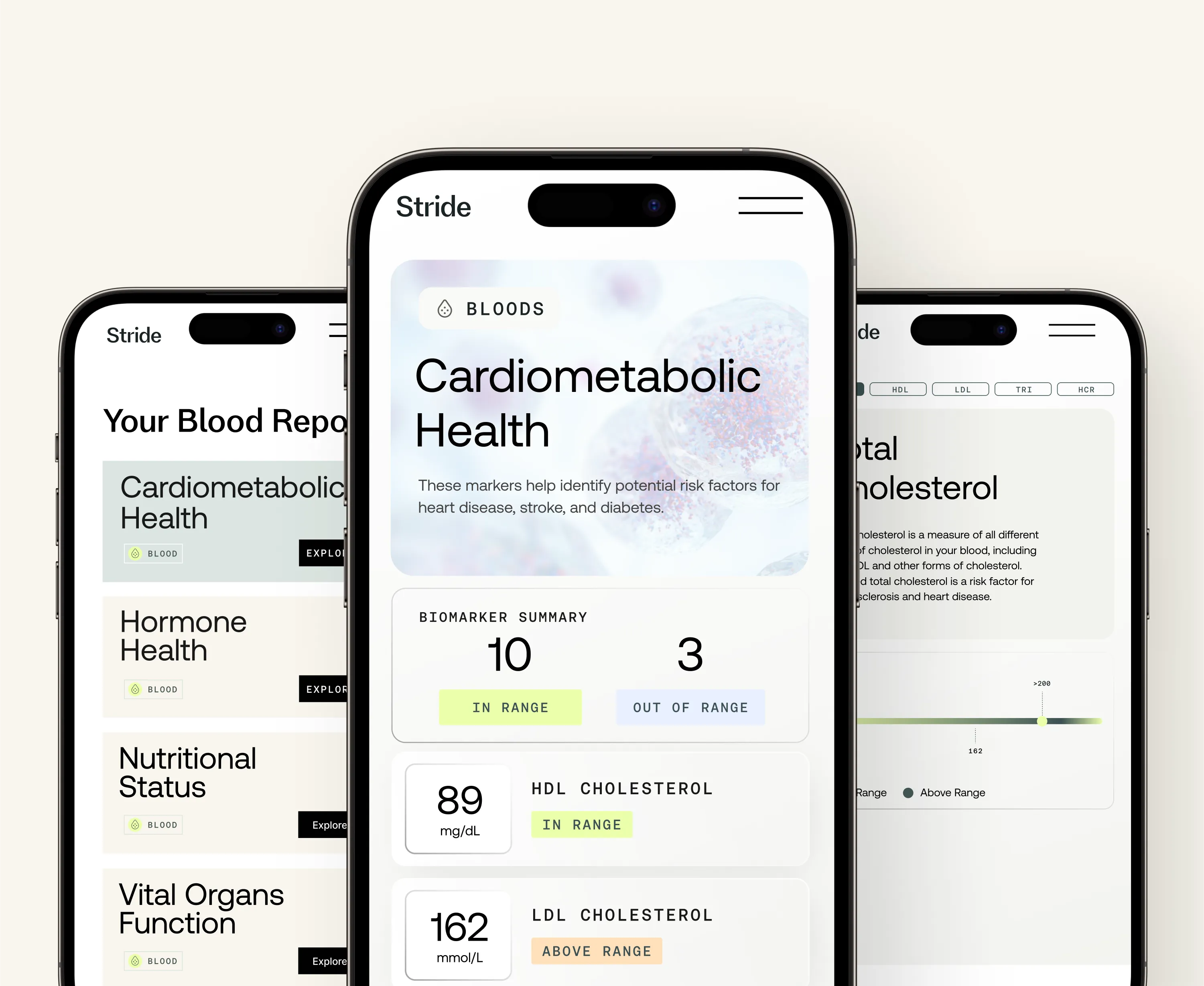
Full blood count identifies anaemia (iron deficiency, B12 deficiency, folate deficiency) which can cause fatigue that accompanies or worsens gut symptoms, and checks for raised white blood cell counts that suggest infection or inflammatory disease.
Coeliac antibodies (tTGA IgA) screen for coeliac disease, which affects approximately 1 per cent of the UK population and produces symptoms indistinguishable from IBS. Coeliac testing must be done while gluten is being consumed: removing gluten before testing renders the antibody test unreliable.
Thyroid function (TSH, Free T4, Free T3) identifies hypothyroidism, which slows gut motility and can cause or worsen constipation-predominant symptoms in ways that dietary intervention alone will not resolve.
CRP (C-reactive protein) and calprotectin distinguish functional gut disorders from inflammatory ones. Elevated CRP or calprotectin suggests gut wall inflammation that warrants further investigation for IBD or other structural conditions.
Ferritin, B12, and vitamin D assess nutritional status, which is commonly compromised in people with long-standing gut symptoms due to impaired absorption, altered dietary intake, and microbiome-related nutrient metabolism changes.
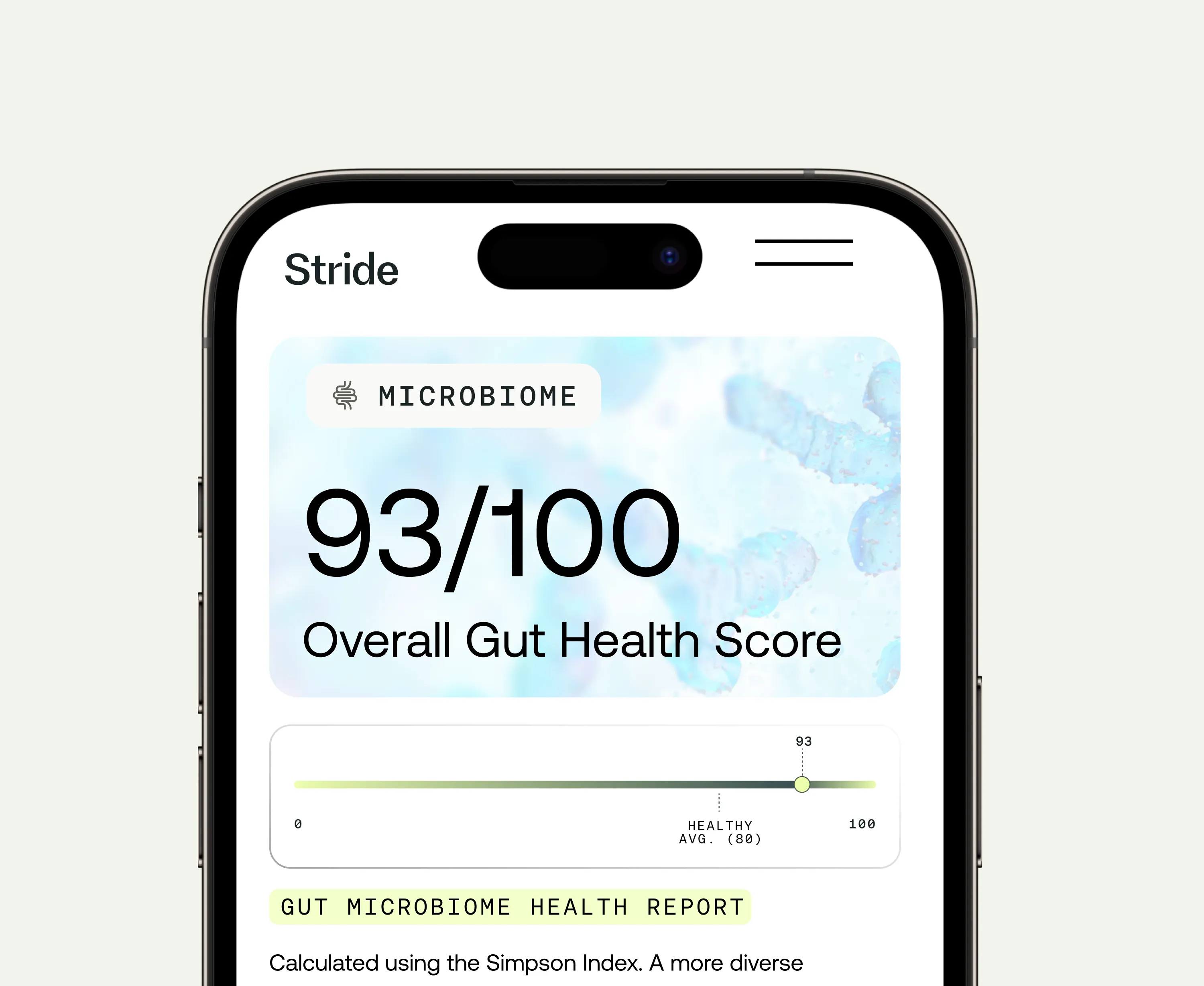
Gut microbiome analysis using DNA sequencing of a stool sample is the only investigation that directly assesses the bacterial composition driving functional symptoms. In people with confirmed IBS, microbiome testing can identify specific dysbiotic patterns, guide targeted probiotic and dietary interventions, and provide a baseline for tracking whether changes are producing measurable improvement in microbiome health over time.
Evidence-based strategies for managing IBS symptoms
Low-FODMAP dietary approach (short-term)
The low-FODMAP diet involves temporary elimination of highly fermentable carbohydrates, followed by systematic reintroduction to identify individual triggers. It is supported by strong clinical trial evidence, with approximately 70 per cent of IBS patients reporting significant symptom improvement during the elimination phase. It is designed as a short-term diagnostic tool (two to six weeks of elimination, then structured reintroduction), not a permanent dietary pattern: long-term restriction of FODMAPs reduces bacterial diversity and is not recommended indefinitely. Working with a dietitian experienced in the low-FODMAP approach maximises outcomes and prevents unnecessary long-term restriction.
Gut microbiome-targeted dietary changes
Building dietary diversity (30 or more plant foods per week), increasing soluble fibre (oats, psyllium husk, legumes), and adding fermented foods supports the microbiome shifts most beneficial for IBS. Unlike the low-FODMAP approach, which is symptom-focused, microbiome-targeted nutrition is mechanism-focused: it directly addresses the dysbiosis patterns that clinical research identifies as drivers of IBS-associated symptoms. Knowing your specific microbiome composition allows these general principles to be personalised to your bacterial profile.
Targeted probiotic supplementation
Specific probiotic strains have clinical evidence for IBS symptom reduction, including Lactobacillus rhamnosus GG, Bifidobacterium infantis 35624, and multi-strain formulations containing Lactobacillus and Bifidobacterium species. A 2024 meta-analysis of 20 randomised controlled trials involving more than 3,000 patients confirmed that probiotics outperformed placebo for overall IBS symptom improvement, abdominal pain reduction, and quality of life. The evidence is stronger for shorter treatment durations (four to eight weeks) and multi-strain formulas, and varies significantly by IBS subtype, which is why knowing your microbiome profile can inform which specific strains to prioritise.
Stress management and gut-brain axis interventions
Interventions with specific evidence for IBS management through the gut-brain axis include cognitive behavioural therapy (CBT), gut-directed hypnotherapy (which has NICE approval for IBS), mindfulness-based stress reduction, and regular aerobic exercise. These are not peripheral adjuncts to IBS management but mechanistically direct interventions that reduce visceral hypersensitivity, lower cortisol, improve vagal tone, and shift the microbiome toward less inflammatory compositions.
Stride tests that can help with IBS symptoms
Health Tests
Optimal Biome
Gut microbiome test for comprehensive analysis & personalized health solutions
From £399 £279.30
Health Tests
Optimal Bloods
At-home test for more than 70 blood-based biomarkers
From £399 £279.30
Biomarkers
| Biomarker | What it measures | Why it matters | Relevance |
|---|---|---|---|
| TSH Blood Test (Thyroid Stimulating Hormone) | Thyroid stimulating hormone | Hypothyroidism slows gut motility; commonly missed driver of constipation-predominant symptoms | 4 |
| Ferritin Blood Test | Iron storage | Iron deficiency anaemia produces fatigue alongside gut symptoms; impaired iron absorption is common in gut conditions | 4 |
| hsCRP Blood Test (High Sensitivity C-Reactive Protein) | Systemic inflammation | Elevated CRP in the context of gut symptoms warrants investigation for IBD; low CRP supports functional diagnosis | 4 |
| Active B12 Blood Test (Holotranscobalamin) | B12 status | Low B12 is common with gut malabsorption; contributes to fatigue and neurological symptoms alongside gut issues | 3 |
| Vitamin D Blood Test (25-OH) | 25-OH vitamin D status | Deficiency is associated with gut wall inflammation and impaired immune regulation in the intestinal mucosa | 3 |
FAQs
What are the main symptoms of IBS and how do I know if I have it?
IBS is characterised by recurrent abdominal pain or cramping alongside altered bowel habits: constipation (IBS-C), diarrhoea (IBS-D), or a mix of both (IBS-M). Pain typically improves with defecation and worsens with certain foods or stress. Bloating is extremely common, often worse as the day progresses. Symptoms vary between people and fluctuate over time, with periods of relative calm interrupted by flare-ups. An IBS diagnosis requires these symptoms to have been present at least once per week for three months (Rome IV criteria). Importantly, IBS should be confirmed by a GP who has excluded other diagnoses including coeliac disease, IBD, and thyroid dysfunction, as these conditions can produce identical symptoms.
What blood tests should be done to rule out other causes of IBS symptoms?
The standard blood tests recommended before accepting an IBS diagnosis include: full blood count (to identify anaemia), coeliac antibodies (tTGA IgA, to screen for coeliac disease), CRP (to exclude significant gut inflammation), thyroid function (TSH), ferritin, vitamin B12, and fasting glucose. Coeliac testing must be performed while gluten is still being consumed regularly: removing gluten before testing produces a false negative. Depending on the specific symptom profile, additional tests including liver function, calprotectin (a gut-specific inflammation marker), and inflammatory bowel disease markers may be appropriate. A comprehensive blood panel performed before or alongside gut microbiome testing gives the most complete picture.
Can a gut microbiome test diagnose IBS?
No single test, including a gut microbiome test, currently diagnoses IBS. IBS remains a clinical diagnosis based on symptoms after exclusion of other conditions. However, gut microbiome testing does provide meaningful information for people with IBS by identifying the specific dysbiotic patterns most associated with their symptom profile, guiding targeted probiotic and dietary interventions, and providing a baseline to track whether interventions are producing measurable microbiome changes. Research is identifying increasingly specific IBS-associated microbial signatures that may eventually inform diagnostic and treatment approaches, but at present microbiome testing is most useful as a management tool rather than a diagnostic one.
Is IBS related to gut bacteria and the microbiome?
Yes, and the research base has strengthened considerably. Studies consistently find that people with IBS have reduced gut bacterial diversity and specific alterations in microbiome composition compared to healthy controls, including lower butyrate-producing bacteria (Faecalibacterium prausnitzii, Roseburia) and altered ratios of major bacterial phyla. A 2024 cross-cohort metagenomic analysis of more than 9,000 samples identified a reproducible IBS-associated microbial signature across different geographic populations for the first time. The microbiome influences IBS through multiple mechanisms: gut barrier integrity, visceral sensitivity, serotonin production, immune activation, and the fermentation patterns that produce gas and alter gut motility.
What is the difference between IBS and IBD?
IBS (irritable bowel syndrome) is a functional gut disorder: the gut appears structurally normal on imaging and endoscopy, but its function is altered. IBD (inflammatory bowel disease) encompasses Crohn's disease and ulcerative colitis, which involve actual inflammation and tissue damage to the gut wall, visible on endoscopy and associated with elevated inflammatory markers. The distinction matters enormously because IBD requires specific medical treatment including immunosuppressive medications, while IBS is managed through diet, lifestyle, and symptom-targeted interventions. Calprotectin (a stool marker of gut wall inflammation) and CRP (a blood inflammation marker) are key tests for distinguishing functional from inflammatory disease. Normal calprotectin strongly supports an IBS diagnosis; elevated calprotectin warrants colonoscopy to investigate IBD.
How does stress make IBS worse?
Stress triggers IBS symptom flares through multiple mechanisms. Acute stress activates the hypothalamic-pituitary-adrenal (HPA) axis, releasing cortisol, which alters gut motility, increases gut permeability, and shifts the microbiome composition toward more dysbiotic patterns. The enteric nervous system (ENS), which governs gut function, is directly modulated by central nervous system stress signals via the vagus nerve. People with IBS have enhanced visceral hypersensitivity (a lower pain threshold in the gut) that is further amplified by stress, explaining why psychological stressors trigger not just emotional distress but genuine physical gut symptoms. Managing the stress response through exercise, mindfulness, CBT, or gut-directed hypnotherapy addresses a mechanistically direct driver of IBS rather than simply treating the psychological component in isolation.
Can a low-FODMAP diet cure IBS?
The low-FODMAP diet is not a cure for IBS but is an effective symptom management strategy for approximately 70 per cent of people who follow it correctly. It works by temporarily reducing the fermentable carbohydrates that gut bacteria rapidly ferment to produce gas, bloating, and altered motility. The critical aspect is that the elimination phase is followed by systematic reintroduction to identify individual trigger foods, not permanent restriction: long-term low-FODMAP eating reduces gut bacterial diversity and can worsen the underlying dysbiosis that contributes to IBS. A dietitian experienced in the low-FODMAP approach will guide this process. The diet manages symptoms but does not address the underlying microbiome patterns, which is why combining it with microbiome-targeted interventions produces more durable outcomes.
Is IBS more common in women than men?
Yes. IBS affects women approximately twice as often as men in Western populations. The reasons include sex differences in gut motility regulation (oestrogen and progesterone directly influence gut transit time and visceral sensitivity), differences in gut microbiome composition between sexes, higher rates of anxiety and depression in women (which bidirectionally worsen IBS), and differences in pain processing and visceral hypersensitivity. Perimenopause and menopause often trigger or worsen IBS symptoms as oestrogen levels change. Women with IBS-C predominant symptoms often notice that their gut symptoms correlate with their menstrual cycle, reflecting the direct hormonal influence on gut motility.
Can IBS cause nutrient deficiencies?
IBS itself does not impair nutrient absorption (unlike coeliac disease or IBD), but the restrictive dietary patterns that many people adopt to manage IBS can lead to nutritional inadequacy. Long-term low-FODMAP dieting without reintroduction can reduce fibre intake significantly and deplete gut bacterial diversity. Avoiding dairy products can lower calcium and iodine intake. Avoiding gluten-containing grains without medical reason (in people who do not have coeliac disease) may reduce B vitamin intake. Additionally, if an underlying condition such as coeliac disease or IBD is the actual cause of symptoms, nutrient malabsorption can be substantial. Testing ferritin, B12, vitamin D, and folate alongside gut investigations ensures nutritional status is assessed as part of the overall picture.
Find out what your body is actually trying to tell you.
Answer 8 quick questions and get a free personalised report - covering which area of your biology is most worth looking at first, what testing in that area reveals, and what actions become available once you have that data.
Where do you feel you're operating below your potential right now?
Methylation & Cellular Health
Your biology is signalling a methylation upgrade - here’s what that means and what to do about it.










