ADHD and nutrition: what the research says about biomarkers and diet
ADHD and nutrition research has expanded significantly in recent years, and the relationship between specific nutrient levels, dietary patterns, and ADHD symptom severity is now supported by a growing body of peer-reviewed evidence. ADHD (attention deficit hyperactivity disorder) was the second most viewed health condition on NHS resources in 2024, reflecting the scale of public interest in understanding a condition that affects an estimated 2.5 million adults in the UK. This page does not address the diagnosis or medication treatment of ADHD, which require clinical assessment. What it does address is the nutritional layer: the biomarkers that are consistently found to be lower in people with ADHD than in matched controls, the dietary patterns associated with better attention and behaviour, and the case for investigating these factors alongside, not instead of, any existing clinical care.
What does the research say about biomarkers and ADHD?
Iron and ferritin
Iron is required for the synthesis of dopamine and noradrenaline, the neurotransmitters most directly involved in attention, impulse regulation, and executive function. Research consistently finds lower serum ferritin levels in people with ADHD compared to healthy controls, with several studies reporting that ferritin levels were approximately twice as low in the ADHD group. Low serum ferritin is inversely related to ADHD severity: the lower the ferritin, the more pronounced the symptom burden. A systematic review of 20 case-control studies found that three studies investigating brain iron specifically (using neuroimaging) found significantly reduced iron in the thalamus of children with ADHD, a region central to attention regulation.
Notably, children with iron deficiency in infancy (at 12 and 18 months) showed more frequent symptoms of ADHD-type inattention and cognitive slowing at ages 5, 10, and 16, suggesting that adequate iron in early development has long-term bearing on attentional capacity. For adults with ADHD, checking ferritin provides an accessible, low-cost starting point for nutritional investigation. Restless leg syndrome, which frequently co-occurs with ADHD, is also strongly associated with low ferritin, providing an additional reason to investigate iron stores.
Omega-3 fatty acids
The brain is approximately 60% lipid by dry weight, with DHA (docosahexaenoic acid) making up 20 to 25% of the fatty acid content of neuronal membranes. Lower omega-3 levels are consistently reported in children and adults with ADHD compared to controls. A systematic review and meta-analysis found that omega-3 supplementation improved clinical and cognitive outcomes in children and adolescents with ADHD, with the most consistent effects observed in people with confirmed low baseline levels. Studies have found a negative correlation between omega-3 index scores and ADHD-related learning and language disorder scores, meaning that the lower the omega-3 status, the greater the symptom severity in these domains.
Dietary sources of EPA and DHA include oily fish (salmon, mackerel, sardines), with algae-based supplements providing a plant-based alternative. The omega-3 to omega-6 ratio in modern diets, heavily weighted toward omega-6 through vegetable oils and processed food, may be as relevant as absolute omega-3 levels.
Magnesium
A 2019 meta-analysis of seven studies found that individuals with ADHD had significantly lower serum magnesium levels than healthy controls. A more recent 2024 review confirmed that lower hair and serum magnesium is consistently seen in ADHD populations. Research has found a moderate, negative correlation between RBC magnesium levels and ADHD symptom severity: the lower the magnesium, the higher the disruptive behaviour and overall symptom scores. Magnesium's role in regulating neurotransmitters and supporting the formation of new neural connections makes it biologically plausible as a contributor to attentional difficulties. Clinical trials in children with both ADHD and magnesium deficiency have shown improvements in hyperactivity and inattention after six months of supplementation.
Zinc
Zinc plays a direct role in dopamine metabolism and receptor function. Case-control studies consistently find lower serum zinc in children with ADHD compared to controls. Low zinc levels in ADHD are specifically associated with inattention scores; the relationship with hyperactivity is less consistent. A dose-response meta-analysis of six randomised controlled trials found that zinc supplementation produced statistically significant improvements in total ADHD scores, with greater effects after longer supplementation periods.
Vitamin D
Research consistently finds lower vitamin D levels in children with ADHD than in age-matched controls. A study of 90 children found that those with ADHD had markedly lower vitamin D levels than those without, and that vitamin D supplementation produced measurable improvements in ADHD symptoms including attention and behavioural regulation. Vitamin D receptors are present throughout the brain, and vitamin D is involved in the regulation of neurotrophic factors and neurotransmitter systems relevant to attention. In the UK, where sunlight-driven synthesis is limited for much of the year, low vitamin D is extremely prevalent.
Vitamin B12, folate, and methylation
Research in 2024 (Neumann et al.) found that lower methylation status at birth was associated with later development of ADHD symptoms, and that DNA methylation may influence ADHD via neurotransmitter functioning and neurite outgrowth. Both B12 and folate are essential co-factors in the methylation cycle, and omega-3 fatty acids have been found to decrease DNA methylation and support neurite outgrowth, linking nutritional status to epigenetic mechanisms relevant to ADHD. A study analysing serum biomarkers in children with ADHD found that homocysteine (a marker of B12, B6, and folate sufficiency) was significantly elevated compared to healthy controls, suggesting impaired methylation in this population.
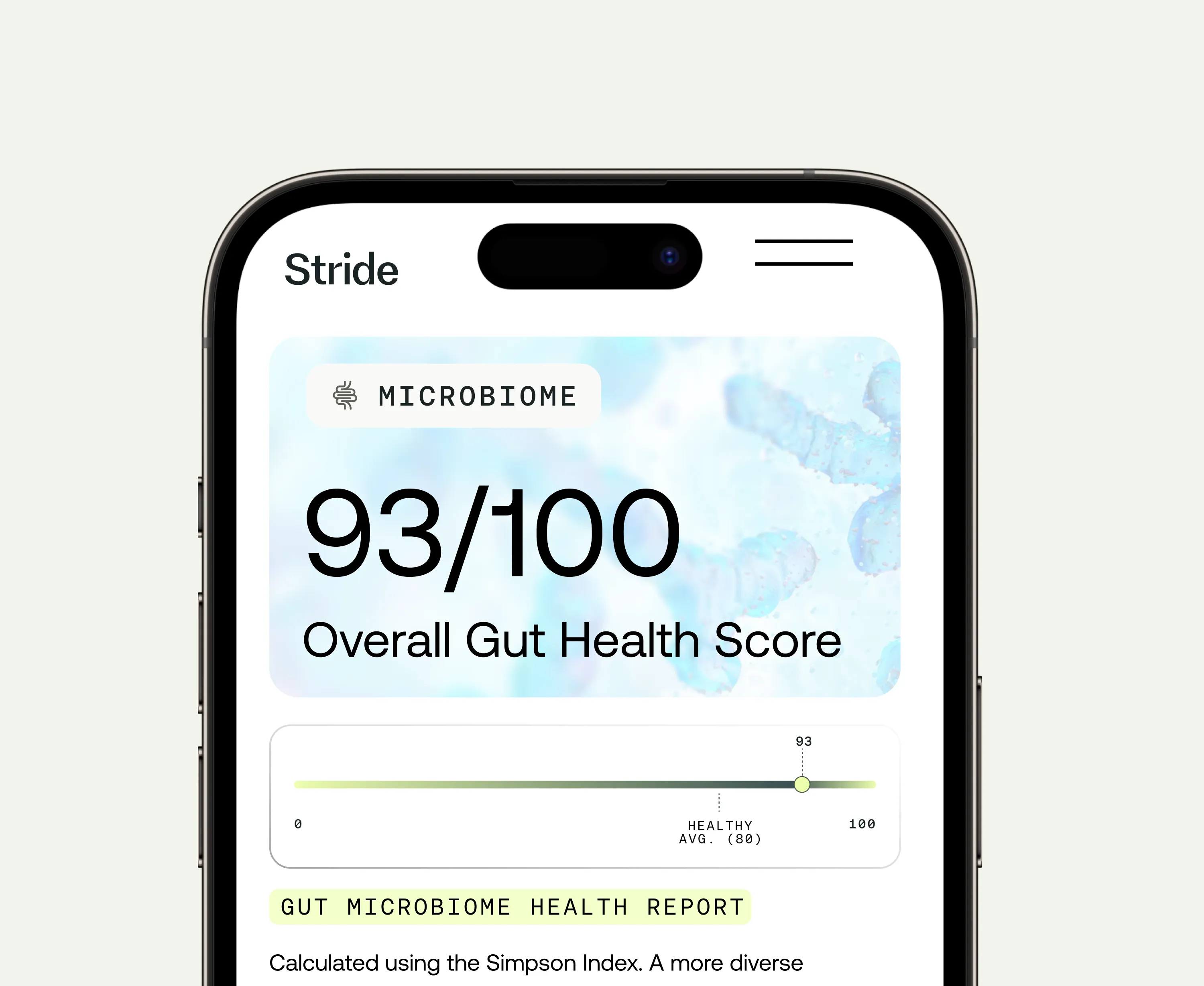
Gut microbiome
Emerging research is investigating the gut-brain axis as a relevant pathway in ADHD. Gut dysbiosis affects systemic inflammation, neurotransmitter availability, and nutrient absorption. Clinical cohort research found that over 80% of children with ADHD and neurodivergence showed high reactivity scores to cow's milk, other dairy, and casein, and over half showed reactivity to wheat and gluten, suggesting that gut-mediated inflammatory responses may be a contributing factor in a subset of people. Microbiome testing provides a baseline assessment of gut diversity and specific imbalances that may be driving systemic effects.
How to investigate nutritional contributors to ADHD
This section addresses testing for the nutritional and biochemical factors associated with ADHD symptom severity. It is not a diagnostic investigation for ADHD itself, which requires clinical assessment by a qualified professional.
A comprehensive blood panel for investigating nutritional contributors to ADHD covers:
Ferritin is the single most consistently implicated blood marker in ADHD research and the most practical starting point. A standard blood count does not assess ferritin; it needs to be specifically requested.
Vitamin D is frequently low in ADHD populations and in the UK population generally. Testing provides a clear baseline for supplementation decisions.
Vitamin B12 and folate assess methylation capacity, which is increasingly implicated in ADHD neurobiology. Homocysteine provides an integrated picture of whether the methylation pathway is functioning adequately.
HbA1c and fasting glucose provide context for blood sugar regulation, which affects attention and cognitive performance throughout the day.
CRP identifies systemic inflammation, which is independently associated with attentional difficulties and may reflect gut-mediated inflammatory processes.
Dietary patterns and ADHD: what the research shows
The Mediterranean dietary pattern
Research consistently finds that lower adherence to a Mediterranean dietary pattern is associated with higher rates of ADHD diagnosis. The Mediterranean diet emphasises vegetables, fruits, legumes, whole grains, and oily fish, precisely the foods richest in the nutrients most consistently implicated in ADHD: iron, zinc, magnesium, omega-3 fatty acids, B vitamins, and vitamin D. Conversely, Western dietary patterns, heavy in ultra-processed food, refined carbohydrates, and sugar, are associated with higher ADHD symptom burden. A systematic review and meta-analysis found that higher junk food consumption was significantly associated with ADHD symptoms in children and adolescents.
The practical implication is not that diet causes or cures ADHD, but that the nutritional composition of the diet has measurable effects on the neurotransmitter systems and metabolic processes most relevant to attention and behaviour. Tracking biomarkers before and after dietary change is the most objective way to know whether the change is moving the needle.
Protein and blood sugar regulation
Protein provides tyrosine, the amino acid precursor to dopamine and noradrenaline. Ensuring adequate protein at each meal, particularly at breakfast, supports neurotransmitter synthesis through the day. Blood sugar instability, common in people who skip meals or eat high-carbohydrate, low-protein patterns, produces attentional dips and emotional dysregulation that are distinct from ADHD but compound its effects. Eating protein with each meal, choosing slower-release carbohydrates, and avoiding long gaps between meals supports more consistent cognitive function.
Reducing food reactivity
For people with suspected gut-mediated food reactivity, an elimination approach followed by structured reintroduction is more informative than blanket dietary restriction. Gut microbiome testing can identify dysbiosis patterns that suggest whether food reactivity is driven by an imbalanced microbial community rather than true allergy. This approach is most relevant for people whose ADHD symptoms fluctuate significantly around specific foods or show a strong gut-symptom relationship.
Tracking changes over time
Because ADHD symptoms fluctuate with context, sleep, and stress, self-reported symptom improvement is a less reliable measure of nutritional impact than biomarker change. Checking ferritin, vitamin D, B12, and CRP at baseline and then retesting after three months of dietary change or supplementation tells you whether the biological markers have shifted. Clinical improvement often follows biochemical improvement with a lag, so biomarker tracking is the more sensitive early indicator.
Stride tests that can help with ADHD
Health Tests
Optimal Biome
Gut microbiome test for comprehensive analysis & personalized health solutions
From $539 $377
Biomarkers
| Biomarker | What it measures | Why it matters | Relevance |
|---|---|---|---|
| Ferritin Blood Test | Iron storage levels | Most consistently implicated blood marker in ADHD research; inversely related to symptom severity | 5 |
| Vitamin D Blood Test (25-OH) | 25-OH vitamin D status | Consistently lower in ADHD populations; supplementation shows symptom benefits in vitamin D-deficient individuals | 5 |
| Active B12 Blood Test (Holotranscobalamin) | Active B12 status | Required for methylation; B12 insufficiency associated with elevated homocysteine in ADHD cohorts | 4 |
| Folate (Vitamin B9) Blood Test | Vitamin B9 status | Co-factor in methylation cycle; relevant to neurotransmitter synthesis and neurodevelopment | 4 |
| hsCRP Blood Test (High Sensitivity C-Reactive Protein) | Systemic inflammation | Chronic inflammation is associated with attentional difficulties; may reflect gut-mediated inflammatory processes | 3 |
| HbA1c Blood Test (Glycated Haemoglobin) | Long-term blood sugar control | Blood sugar instability compounds attentional difficulties; metabolic dysregulation is more common in ADHD populations | 3 |
| TSH Blood Test (Thyroid Stimulating Hormone) | Thyroid function | Thyroid dysfunction can produce attentional symptoms; thyroid hormones are relevant to brain energy metabolism | 2 |
FAQs
Can a blood test help with ADHD?
Blood tests cannot diagnose ADHD. What they can identify is whether measurable nutritional deficiencies or imbalances are contributing to the symptom severity experienced by someone who has ADHD or who suspects they may. Ferritin, vitamin D, B12, folate, and homocysteine are the most consistently relevant markers based on the research. Identifying and correcting a deficiency does not replace medication or behavioural support; it addresses a biological layer that may be making symptoms harder to manage.
Does iron deficiency make ADHD worse?
Research suggests it can. Ferritin levels are consistently lower in people with ADHD than in matched controls, and the severity of iron deficiency correlates with the severity of ADHD symptoms. Iron is required for the synthesis of dopamine and noradrenaline, and brain iron appears to be particularly relevant: neuroimaging studies have found reduced iron in the thalamus of children with ADHD. Low ferritin also causes restless leg syndrome and disrupted sleep, both of which independently worsen attention and impulse control. Checking ferritin is the most practical first step in a nutritional investigation.
Can omega-3 fatty acids help with ADHD?
The evidence supports a benefit specifically in people with documented low omega-3 status. Lower omega-3 levels are consistently found in people with ADHD, and meta-analyses of supplementation trials find improvements in clinical and cognitive outcomes, particularly attention and learning-related scores. The brain's neuronal membranes are 20 to 25% DHA by fatty acid composition, making omega-3 availability directly relevant to the structural biology of attention networks. Dietary sources include oily fish (salmon, mackerel, sardines); algae-based supplements provide a plant-based DHA source.
What is the link between ADHD and vitamin D?
Children with ADHD consistently show lower vitamin D levels than age-matched controls, and supplementation trials in vitamin D-deficient children with ADHD have found improvements in attention, behavioural regulation, and emotional control. Vitamin D receptors are found throughout the brain in regions involved in dopamine regulation, and vitamin D supports the neurotrophic factors that maintain neural connectivity. In the UK, where dietary vitamin D intake is generally low and sunlight synthesis is limited to approximately April to September, deficiency is common and the gap between ADHD and non-ADHD vitamin D levels may partly reflect this population-wide pattern.
Can diet improve ADHD symptoms?
Research suggests that dietary patterns, particularly the Mediterranean diet, are associated with lower ADHD symptom burden, while Western dietary patterns are associated with higher symptom severity. The likely mechanism is the nutritional composition of these patterns rather than any single food: the Mediterranean diet is richer in the micronutrients most consistently implicated in ADHD, including iron, zinc, magnesium, omega-3 fatty acids, and B vitamins. Diet is not a replacement for clinical ADHD treatment, but it is a modifiable factor that can influence the biological environment in which ADHD symptoms are expressed.
What is the relationship between ADHD and gut health?
Emerging research suggests that the gut-brain axis is relevant to ADHD. The gut microbiome influences systemic inflammation, neurotransmitter availability, and nutrient absorption, all of which affect attentional and behavioural function. Clinical cohort studies have found high rates of food reactivity in people with ADHD and neurodivergence, and gut dysbiosis may contribute to the systemic inflammatory patterns associated with ADHD. This is an active area of research rather than an established mechanism, but for people whose ADHD symptoms have a strong gut-symptoms relationship, microbiome investigation may provide useful information.
Can ADHD be related to blood sugar problems?
Blood sugar instability does not cause ADHD, but it can significantly compound attentional difficulties. When blood glucose falls sharply, cognitive performance drops, impulse control worsens, and emotional regulation becomes harder. These effects are distinct from ADHD but are indistinguishable in the moment from symptom worsening. People with ADHD often have dietary patterns that promote blood sugar variability (skipping meals, high refined carbohydrate intake), making metabolic investigation a practical component of a comprehensive nutritional assessment. HbA1c provides a three-month average that tells you whether the overall pattern is one of stable or variable glucose regulation.
Is there a specific ADHD blood test UK available?
There is no blood test that diagnoses ADHD. ADHD is a clinical diagnosis based on symptom history, duration, and functional impairment, assessed by a qualified clinician. What exists is testing for the nutritional and biochemical factors most consistently associated with ADHD symptom severity: ferritin, vitamin D, B12, folate, homocysteine, CRP, and HbA1c. These markers are available as part of a comprehensive blood panel such as Stride Optimal Bloods and can be done at home with a finger-prick or venous draw.
Find out what your body is actually trying to tell you.
Answer 8 quick questions and get a free personalised report - covering which area of your biology is most worth looking at first, what testing in that area reveals, and what actions become available once you have that data.
Where do you feel you're operating below your potential right now?
Methylation & Cellular Health
Your biology is signalling a methylation upgrade - here’s what that means and what to do about it.










