Gut health problems: why your digestion is off and how to find out
Gut health problems affect a significant portion of adults in the UK, with surveys suggesting that more than half of people experience digestive issues in any given year. Yet persistent gut symptoms are routinely dismissed as stress, IBS, or "just the way you are" without investigating the underlying biology. Your gut is home to an estimated 100 trillion microorganisms, collectively known as the gut microbiome, which play a central role in digestion, immune function, mood regulation, inflammation, and metabolic health. When this ecosystem becomes imbalanced (a state called dysbiosis), the consequences extend far beyond bloating and irregular bowel movements. Understanding what is happening in your gut specifically requires looking at the composition and function of your microbiome, and this is where targeted testing becomes useful.
What causes gut health problems?
Dysbiosis: an imbalance in the gut microbiome
Dysbiosis describes a disruption of the normal microbial balance in the gut, characterised by reduced diversity, overgrowth of potentially harmful species, and loss of beneficial bacteria that produce key metabolites including short-chain fatty acids (SCFAs) such as butyrate. Butyrate is the primary fuel source for colonocytes (the cells lining the colon) and plays a direct role in maintaining gut barrier integrity, suppressing inflammation, and regulating immune responses. When butyrate-producing bacteria such as Faecalibacterium prausnitzii and Roseburia decline, the downstream effects ripple across multiple systems far beyond digestion.
Ultra-processed diet and low fibre intake
The single most impactful driver of gut dysbiosis at a population level is dietary fibre insufficiency combined with high ultra-processed food consumption. Gut bacteria feed primarily on dietary fibre (particularly fermentable polysaccharides), producing SCFAs as a byproduct. Most UK adults consume well below the recommended 30g of fibre per day. Ultra-processed foods contain emulsifiers, artificial sweeteners, and preservatives that directly alter gut bacterial composition, reduce diversity, and disrupt the mucus layer protecting the gut lining. Research consistently shows that people consuming 30 or more different plant foods per week have significantly higher gut microbiome diversity than those eating a narrower range.
Antibiotic use
Antibiotics are the most potent known disruptor of the gut microbiome. A single course can reduce bacterial diversity by 30 to 90 per cent, with some species taking months to years to recover. Repeated antibiotic courses, common in childhood and recurring infections, cumulatively deplete bacterial diversity in ways that can persist into adulthood. The disruption created by antibiotics is also a common trigger for post-antibiotic IBS, food sensitivities, and recurrent digestive problems in people who previously had no gut symptoms.
Chronic stress and the gut-brain axis
The gut and brain are in constant bidirectional communication through the enteric nervous system, vagus nerve, and systemic signalling via gut-derived neurotransmitters. Approximately 90 per cent of the body's serotonin is produced in the gut, and gut bacteria directly influence its production. Chronic psychological stress alters gut motility, increases gut permeability (allowing bacterial compounds to enter circulation), and shifts the microbiome composition toward bacteria associated with inflammation. This explains the common clinical observation that gut symptoms worsen dramatically during periods of sustained stress or psychological difficulty.
Impaired gut barrier integrity (leaky gut)
The gut barrier is a single-cell-thick lining that separates the gut lumen from the bloodstream. When bacterial diversity declines and butyrate production falls, the integrity of tight junction proteins that seal this barrier weakens, allowing bacterial fragments and undigested food particles to enter circulation. This triggers systemic low-grade inflammation and immune activation, which has been linked to conditions including autoimmune disease, skin conditions, joint pain, and brain fog. Zonulin, a protein that regulates tight junction permeability, is elevated in people with increased gut permeability and can be measured in stool or blood.
Sleep disruption
The gut microbiome follows a circadian rhythm, with different bacterial communities becoming more or less active across the day-night cycle. Chronic sleep restriction and irregular sleep timing disrupt this rhythm, shifting the microbiome toward compositions associated with metabolic dysfunction, higher inflammation, and impaired glucose metabolism. People with consistently poor sleep show measurably lower microbiome diversity than those with regular, adequate sleep, independent of diet and other lifestyle factors.
Medications beyond antibiotics
Proton pump inhibitors (PPIs), commonly prescribed for acid reflux, alter the gut's pH environment in ways that reduce diversity in the small intestine and promote overgrowth of species associated with dysbiosis. Non-steroidal anti-inflammatory drugs (NSAIDs) increase gut permeability and disrupt the protective mucus layer. Many other common medications, including some antidepressants, metformin, and certain blood pressure medications, have measurable effects on microbiome composition that are increasingly recognised in the research literature.
How to test for gut health problems
Standard NHS testing for gut symptoms typically involves stool tests to rule out infection or blood, blood tests to exclude coeliac disease, thyroid dysfunction, and anaemia, and in persistent cases a referral for colonoscopy or endoscopy. These investigations are important for excluding serious pathology, but they do not assess the composition or function of the microbiome, which is the primary driver of functional gut symptoms in the majority of people without structural disease.
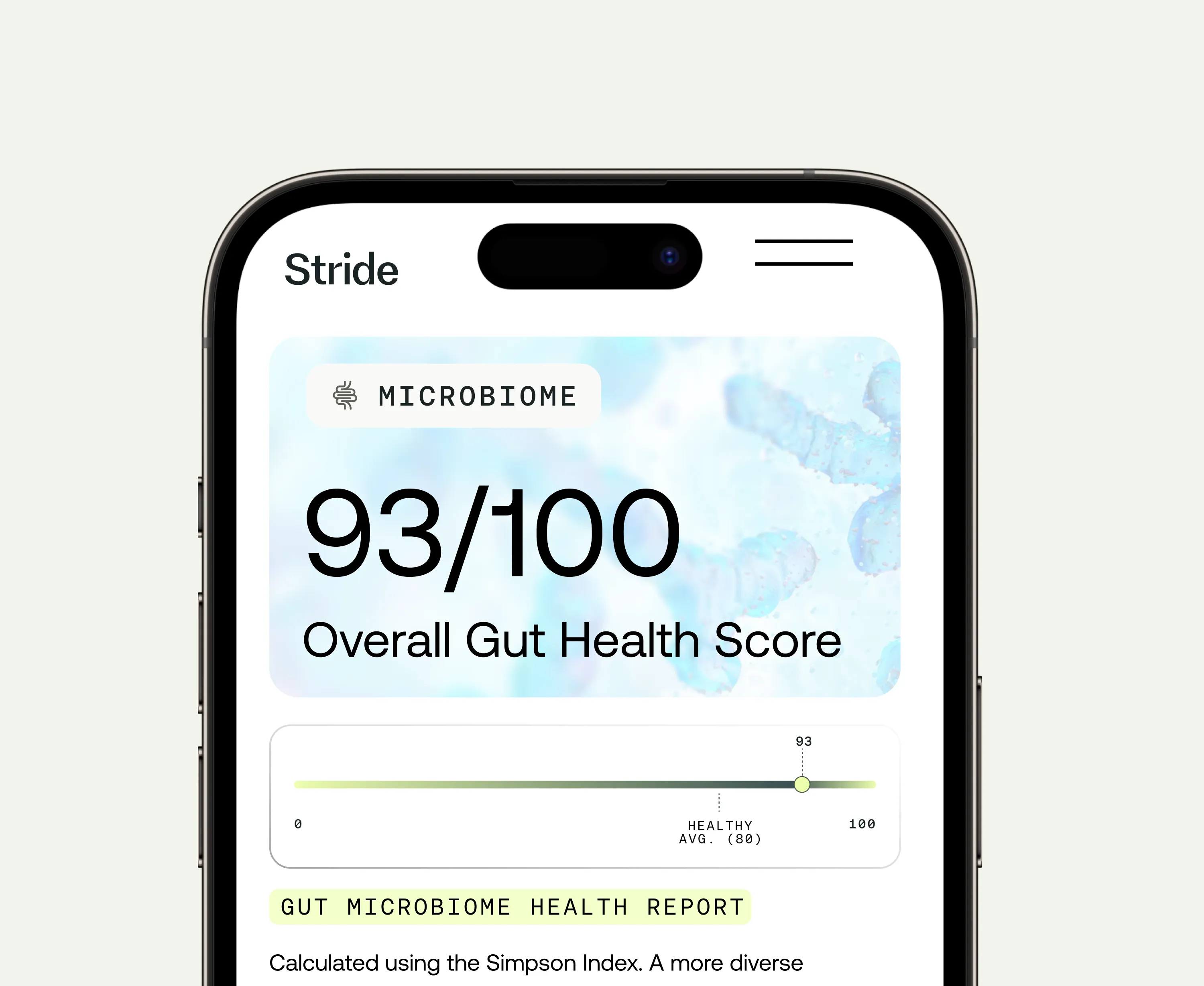
A comprehensive gut microbiome test uses DNA sequencing of a stool sample to identify and quantify the thousands of bacterial species living in your large intestine. Unlike traditional stool cultures (which only grow a limited number of species in the lab), DNA sequencing captures the full microbial picture. The assessment includes:
Bacterial diversity is a primary indicator of gut health. Higher diversity is associated with better metabolic function, more robust immune regulation, and greater resilience to dietary or environmental perturbation. Low diversity is a consistent finding in people with IBS, inflammatory bowel conditions, obesity, type 2 diabetes, and metabolic syndrome.
Beneficial and potentially harmful species are assessed relative to population norms. Key beneficial species include Akkermansia muciniphila (which supports gut barrier integrity and metabolic health), Faecalibacterium prausnitzii (a major butyrate producer and anti-inflammatory species), and Bifidobacterium species (important for SCFA production and immune regulation). Key potentially harmful overgrowths include Clostridium difficile, pathogenic E. coli strains, and Candida yeast species.
Functional markers including short-chain fatty acid production capacity, gut permeability indicators, and inflammatory markers (such as calprotectin) add clinical context to the composition data by showing not just which bacteria are present but what they are producing and what the gut environment looks like.
For people with persistent gut symptoms, combining a microbiome test with a blood panel (checking for coeliac antibodies, thyroid function, anaemia, and inflammatory markers) gives the most complete investigation. Blood markers can identify conditions that cause gut symptoms secondary to another process, while microbiome testing investigates the primary gut ecosystem. Your expert consultation can help determine which combination is most relevant to your symptom profile.
Evidence-based strategies to support gut health
Dietary diversity and the 30-plant rule
Consuming 30 or more different plant foods per week is the single most consistently evidence-supported dietary approach to improving gut microbiome diversity. Plant foods include vegetables, fruits, legumes, whole grains, nuts, seeds, and herbs: variety is more important than quantity, since different bacteria specialise in fermenting different types of fibre and polyphenols. A narrow diet, even a healthy one, can selectively favour certain bacterial communities at the expense of overall diversity.
Fermented foods and live cultures
Regular consumption of fermented foods (natural yogurt with live cultures, kefir, sauerkraut, kimchi, miso, kombucha) has been shown in randomised controlled trials to increase microbiome diversity and reduce inflammatory markers more effectively than a high-fibre diet alone over a six-week period. The mechanism involves both direct introduction of live bacterial cultures and the metabolites produced during fermentation, which create a gut environment that supports existing beneficial species.
Stress management and the vagus nerve
Given the direct bidirectional connection between the gut and the nervous system, stress management interventions with evidence for gut benefit include regular aerobic exercise (which increases vagal tone and directly modulates gut bacterial composition), mindfulness and breathwork (which activate the parasympathetic nervous system and reduce gut motility dysregulation), and adequate sleep. These are not peripheral suggestions but mechanistically direct interventions for the gut-brain axis.
Targeted probiotic and prebiotic supplementation
Not all probiotic supplements have the same evidence base or species composition. Specific strains with meaningful clinical evidence for gut health include Lactobacillus rhamnosus GG (for post-antibiotic recovery and IBS), Bifidobacterium longum (for gut barrier function and anxiety), and Saccharomyces boulardii (a yeast probiotic effective in reducing C. difficile recurrence and traveller's diarrhoea). Prebiotic fibres (inulin, FOS, GOS, resistant starch) feed existing beneficial bacteria and are often more impactful than adding new strains. Knowing your microbiome composition helps identify which targeted interventions are most likely to benefit your specific bacterial profile.
Stride tests that can help with Gut health problems
Health Tests
Optimal Biome
Gut microbiome test for comprehensive analysis & personalized health solutions
From €459 €321
FAQs
What are the signs of poor gut health in adults?
Signs of poor gut health in adults range from the obvious to the less expected. Digestive symptoms include persistent bloating, excess gas, abdominal pain or cramping, irregular bowel movements (constipation, diarrhoea, or alternating between both), and acid reflux. Beyond digestion, poor gut health often manifests as frequent infections and low immune resilience (given that approximately 70 per cent of immune function is located in gut-associated lymphoid tissue), skin conditions including acne, eczema, and rosacea, unexplained fatigue, brain fog, low mood, and anxiety. If you are experiencing a combination of these symptoms across multiple body systems, gut dysbiosis is worth investigating as a potential connecting thread.
What is gut dysbiosis and how does it affect health?
Gut dysbiosis describes an imbalance in the composition and function of the gut microbiome: typically reduced diversity, loss of beneficial species, and relative overgrowth of potentially harmful ones. It is not a disease in itself but a biological state that increases vulnerability to a wide range of conditions. The mechanisms include impaired SCFA production (which reduces gut barrier integrity and anti-inflammatory signalling), increased gut permeability (allowing bacterial fragments to enter circulation and trigger systemic inflammation), disrupted immune regulation, and altered neurotransmitter production via the gut-brain axis. Addressing dysbiosis through dietary changes, targeted supplementation, and lifestyle modification often improves symptoms across multiple apparently unrelated body systems simultaneously.
How does gut health affect mental health and mood?
The gut-brain axis is a bidirectional communication network linking the enteric nervous system in the gut with the central nervous system via the vagus nerve, the immune system, and systemic signalling molecules. Approximately 90 per cent of the body's serotonin is produced in the gut by enterochromaffin cells, and this production is directly influenced by gut bacterial composition. Specific bacteria regulate tryptophan metabolism (the precursor to serotonin), GABA production, and inflammatory signalling that affects brain function. Research consistently shows that people with anxiety and depression have distinct gut microbiome compositions compared to healthy controls, and intervention studies show that microbiome-targeted dietary changes produce measurable improvements in mood and anxiety scores.
Can gut microbiome testing tell me which foods are causing my symptoms?
A gut microbiome test does not identify food intolerances in the way that an IgE allergy test or a coeliac antibody test does. However, it can identify the bacterial patterns most associated with your symptoms and provide personalised dietary recommendations based on which types of fibre and plant foods your microbiome is most suited to ferment. It can also identify overgrowths of bacteria that are particularly reactive to certain fermentable carbohydrates (such as FODMAP-fermenting species), which can explain why low-FODMAP dietary adjustments help some people and not others. For specific food intolerances, targeted blood testing is more appropriate.
What is the difference between the gut microbiome and gut bacteria?
Gut bacteria are one component of the gut microbiome. The microbiome refers to the entire ecosystem of microorganisms living in your digestive tract, which includes bacteria (the most numerous and well-studied), viruses (bacteriophages), fungi (including Candida species), archaea, and other microorganisms. Together these microorganisms and their collective genetic material comprise the microbiome. Current DNA sequencing technology used in consumer gut tests primarily identifies bacterial composition (using 16S rRNA sequencing), though more comprehensive metagenomics approaches also capture fungi and other microorganism types. When people refer to gut health testing, they are almost always referring to the bacterial component.
How long does it take to improve gut health and see changes in symptoms?
Research suggests that measurable changes in gut microbiome composition are detectable within two to four weeks of sustained dietary changes, particularly when fibre diversity and fermented food intake increase significantly. Symptom improvement often follows a similar timeline for functional digestive symptoms such as bloating and irregular bowel movements, though it can take three to six months for more complex patterns to fully resolve. Retesting the microbiome after three to six months of targeted dietary and lifestyle changes is the most objective way to confirm that the interventions are producing the intended microbial shifts for your biology specifically, rather than relying solely on symptom self-reporting.
Is gut microbiome testing available on the NHS?
Standard NHS stool testing screens for infection, blood, and specific pathogens (such as C. difficile), and includes calprotectin testing to identify gut inflammation. Comprehensive gut microbiome analysis using DNA sequencing is not currently available on the NHS as a routine investigation, as clinical interpretation guidelines and standardised reference ranges are still being established through ongoing research. Private microbiome testing provides the most detailed assessment currently available to the general public. The field is evolving rapidly, and gut microbiome profiling is increasingly used in clinical research settings as part of personalised medicine approaches to gastrointestinal and metabolic conditions.
Does poor gut health cause weight gain?
The gut microbiome plays a direct role in energy metabolism and fat storage through multiple mechanisms. The Firmicutes-to-Bacteroidetes ratio in particular has been linked to energy extraction efficiency: a higher ratio is associated with greater calorie extraction from the same diet, contributing to weight gain independent of calorie intake. Low Akkermansia muciniphila levels are associated with higher body weight, increased gut permeability, and metabolic dysfunction. The microbiome also influences appetite-regulating hormones (ghrelin, leptin, GLP-1) and insulin sensitivity. Addressing gut dysbiosis as part of a weight management approach addresses a mechanistic driver that standard calorie-counting does not account for.
Can antibiotics permanently damage the gut microbiome?
Most of the research on antibiotic-related microbiome disruption shows recovery of diversity within one to two months for a single course in otherwise healthy adults, though some species may take considerably longer to return and some individuals show less complete recovery than others. Repeated antibiotic courses over years, particularly in childhood when the microbiome is still establishing its adult composition, can produce more lasting diversity losses. Specific probiotic strains (particularly Lactobacillus rhamnosus GG and Saccharomyces boulardii, taken alongside the antibiotic course and for two to four weeks after) have the strongest evidence for supporting recovery. High dietary fibre diversity during and after a course of antibiotics accelerates the return of diversity significantly.
Find out what your body is actually trying to tell you.
Answer 8 quick questions and get a free personalised report - covering which area of your biology is most worth looking at first, what testing in that area reveals, and what actions become available once you have that data.
Where do you feel you're operating below your potential right now?
Methylation & Cellular Health
Your biology is signalling a methylation upgrade - here’s what that means and what to do about it.










